Please see Indication and Important Safety Information, including Boxed Warning, throughout this video and in Chapter 5.
ON SCREEN: Why Choose BOTOX® for Overactive Bladder?
VO: Treat overactive bladder (OAB) differently. Discover BOTOX®
BOTOX® could be an OAB treatment that you’ve been searching for, when an OAB pill (known as an anticholinergic) hasn't worked well enough or can't be tolerated.
VO: What does living with OAB look like?
The journey for you and your doctor to find an OAB treatment that works well for you can sometimes be a challenge, and you may be wondering if BOTOX® could be an appropriate treatment for you.
ON SCREEN: OAB = overactive bladder.
VO: Living with OAB has a way of redirecting the plans that you’ve made. Maybe OAB keeps you from doing some of the activities you enjoy because of concerns about wetting yourself or frequent bathroom visits, or you feel embarrassed to be around others because they may notice your symptoms, and you may be frustrated, dealing with the other limitations that OAB can impose on your everyday life like deciding what you would like to wear.
VO: There are more than 33 million adults in the US affected by OAB, but everyone deals with it a little differently. There is no one-size-fits-all treatment. That's why you need a personalized approach that works for you.
Now, maybe you’ve tried different OAB treatments and haven’t gotten the results you hoped for. For instance, you’ve taken daily OAB pills that you couldn’t tolerate or didn’t work well enough for you.
ON SCREEN: OAB pills, known as anticholinergics.
VO: Then it may be time to ask your doctor if BOTOX® is right for you.
Please see Important Safety Information, continued within this chapter, and in Chapter 5.
VO: How well does BOTOX® work?
BOTOX® was proven to significantly reduce daily leakage episodes after the first treatment.
In clinical trials, 3 months after treatment, people treated with BOTOX® had an average of about 3 fewer leakage episodes per day, compared to 1 fewer leakage episode per day with placebo.
ON SCREEN: In clinical studies, results were measured at 2, 6, and 12 weeks, with 12 weeks being the primary time point.
VO: You may notice results as soon as 2 weeks after treatment.
ON SCREEN: In clinical studies, results were measured at 2, 6, and 12 weeks, with 12 weeks being the primary time point.
VO: Take a look at the results from an additional measure in the BOTOX® clinical studies.
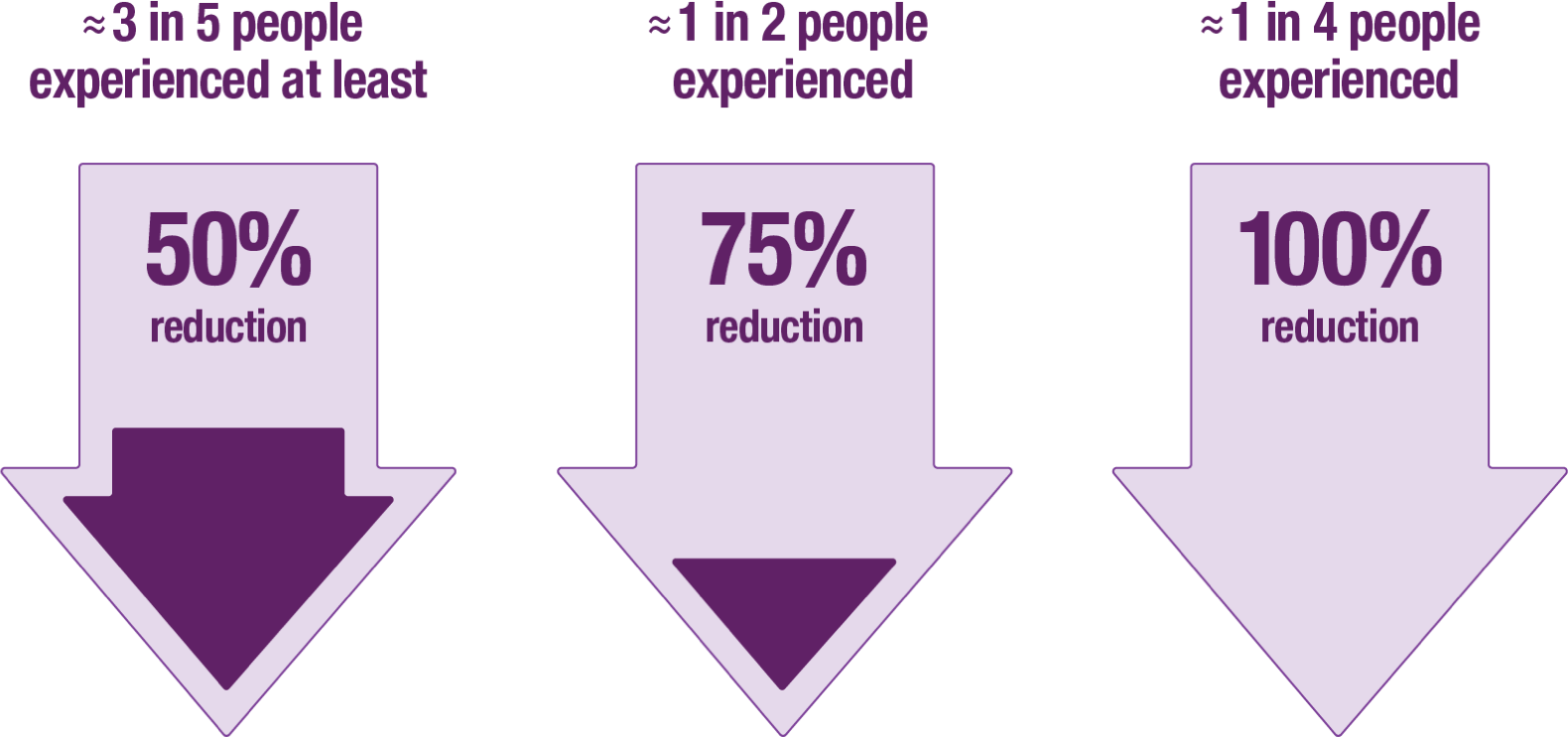
VO: Clinical trials showed that 3 months after treatment, about 3 in 5 people experienced at least 50% reduction in daily leakage episodes, and about 1 in 2 people experienced 75% reduction in daily leakage episodes. Approximately 1 in 4 people reported that their leakage episodes stopped completely. Please review the study-related information and limitation statement shown.
ON SCREEN: People who experienced a reduction in daily leakage episodes with BOTOX®
3 months after treatment in clinical trials*:
≈ 3 in 5 people experienced at least 50% reduction
≈ 1 in 2 people experienced 75% reduction
≈ 1 in 4 people experienced 100% reduction
Limitation: The percentage of patients achieving 50% to 100% reduction in daily leakage episodes shown above was an additional measure in the primary clinical studies.
*These results are based on an analysis of the primary clinical data (reduction in daily leakage episodes). No definitive conclusions about the treatment effects with BOTOX® can be drawn from these results.
Study 1: BOTOX® n = 280, placebo n = 277; Study 2: BOTOX® n = 277, placebo n = 271:
- Patients with ≥ 50% reduction: BOTOX® 57.5% and 63.5%; placebo 28.9% and 33.2%
- Patients with ≥ 75% reduction: BOTOX® 44.6% and 47.3%; placebo 15.2% and 20.3%
- Patients with 100% reduction: BOTOX® 22.9% and 31.4%; placebo 6.5% and 10.3%
VO: When evaluating how well a treatment works for you, it's also important to think about your overall quality of life. In BOTOX® clinical trials, a questionnaire showed patients reported improvement in their overall quality of life based on three categories. Avoiding situations and limiting behaviors, such as overplanning daily details, concern about bathroom accessibility, and limiting how much they drink. Social and psychological impact, including a preoccupation with symptoms in everyday decisions, such as clothing and travel choices, as well as their perceived overall well-being. And social embarrassment, which includes self-consciousness or worry that others may notice their symptoms.
ON SCREEN:
Incontinence Quality of Life (I-QOL) questionnaire
- Avoiding situations and limiting behaviors
- Social and psychological impact
- Social embarrassment
Limitation: I-QOL was an additional measure in the primary clinical studies.
This improvement was reported using the Incontinence Quality of Life questionnaire (I-QOL). This is a validated questionnaire that is used to measure the impact of urinary incontinence on a patient's quality of life.
VO: Are you seeing the results you had hoped for from your OAB pill? If not, talk to your doctor about BOTOX®.
ON SCREEN: OAB pills, known as anticholinergics.
Please see Important Safety Information, continued within this chapter, and in Chapter 5.
VO: How does BOTOX® work?
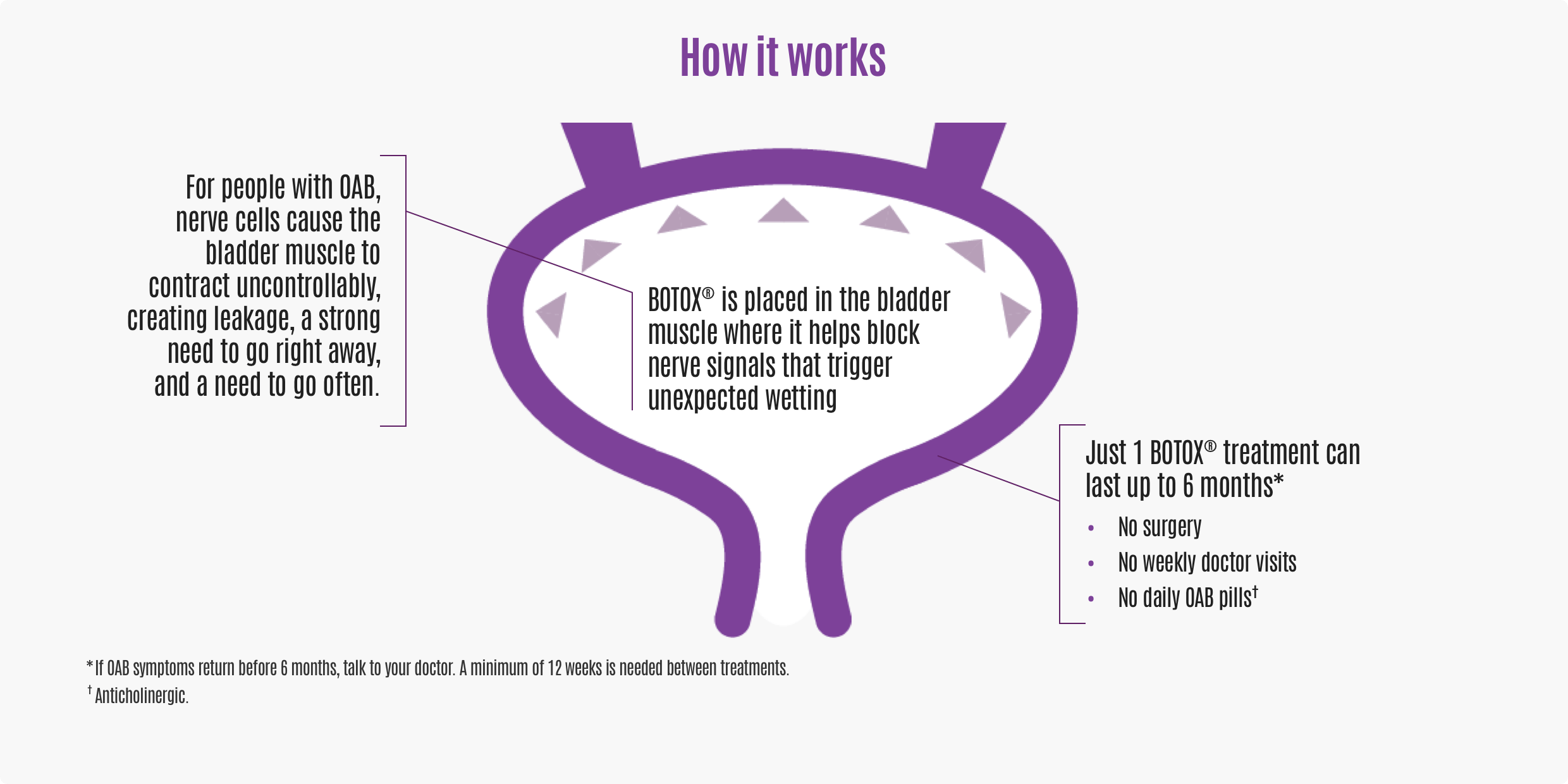
BOTOX® takes a different approach from other medications. It delivers treatment directly where OAB starts—in the bladder—without the need for surgery, weekly doctor visits, or daily OAB pills*.
ON SCREEN: *OAB pills, known as anticholinergics.
VO: For people with OAB, nerve cells cause the bladder muscle to contract uncontrollably, creating leakage, a strong need to go right away, and a need to go often. BOTOX® is placed in the bladder muscle where it helps block nerve signals that trigger unexpected wetting. In fact, just 1 BOTOX® treatment can last up to 6 months.* That means no need for surgery, no weekly doctor visits, and no need for taking daily OAB pills.† How would that change your own OAB treatment journey?
ON SCREEN: *If OAB symptoms return before 6 months, talk to your doctor. A minimum of 12 weeks is needed between treatments.
†OAB pills, known as anticholinergics.
VO: Once you and your doctor have decided that BOTOX® is right for you, what should you expect during your treatment appointment?
VO: The treatment may be given in the convenience of your doctor’s office, and the entire appointment takes about an hour. Your doctor will prescribe you antibiotics to help prevent a urinary tract infection. You will be instructed to take them before, on, and after the day of treatment.
VO: During the first 20 minutes, the staff will help you get settled and comfortable. They will also check for a urinary tract infection. You should not be given BOTOX® if you have a urinary tract infection. Be sure to let them know if you have any questions or concerns about the procedure and if there are ways they can help you feel more comfortable. Next, the doctor will administer some numbing medicine inside your bladder muscle.
VO: Once your bladder is numb, it takes about 10 minutes to administer BOTOX®. A small lighted tube called a cystoscope is inserted into the bladder through the natural opening where urine comes out. This allows the doctor to place BOTOX® within the bladder. After the procedure, your doctor will want you to remain in the office for about 30 minutes to make sure you can easily empty your bladder. During this time, you can schedule your next appointment, and then leave to go about your day.
VO: You should not experience significant pain after treatment, although it may sting or burn when you urinate the first few times. You may also see some blood in the urine right after treatment.
ON SCREEN: Call your doctor if any of these symptoms persist
VO: The most common side effects of BOTOX® in clinical trials for OAB were urinary tract infection, painful or difficult urination, and temporary inability to fully empty your bladder. This is not a complete list of possible side effects. Please see the Important Safety Information, including Boxed Warning, within this video and talk to your doctor about any concerns you may have.
ON SCREEN: Please see the Important Safety Information, including Boxed Warning, within this video and talk to your doctor about any concerns you may have.
VO: Nearly all people could still urinate when they needed to after BOTOX® treatment; however, there have been instances where some people temporarily could not fully empty their bladder on their own. If that happens, you may need to temporarily use a self catheter. Unlike the type usually used in a hospital, this is a small catheter, about the size of a coffee stirrer, that you can easily use by yourself in any restroom.
ON SCREEN: You should not receive BOTOX® if you are not willing (or able) to use a disposable self-catheter if necessary.
VO: You can learn more about the possible side effects at BOTOXforOAB.com and speak with your doctor about any concerns you may have.
VO: If you’re interested in twice-yearly OAB treatment, ask your doctor if BOTOX® may be right for you.
ON SCREEN: If OAB symptoms return before 6 months, talk to your doctor. A minimum of 12 weeks is needed between treatments.
Please see Important Safety Information, continued within this chapter, and in Chapter 5.
VO: How long does BOTOX® treatment last?
One of the challenges of OAB is that it’s a chronic condition, so it requires ongoing care. But, what if you had up to 6 months between OAB treatments? One BOTOX® treatment can last up to 6 months. Your doctor will determine how often you need to get retreated. With two BOTOX® treatments a year, you may be able to be free from daily OAB pills† that aren’t working well enough or can’t be tolerated.
On Screen: If OAB symptoms return before 6 months, talk to your doctor. A minimum of 12 weeks is needed between treatments.
VO: Staying on a consistent retreatment schedule is important. It’s about every 6 months and can be as simple as setting your next appointment before leaving the office after your treatment. That way you’re already on the books, and you can easily adjust it around your schedule. If your symptoms return before 6 months, talk to your doctor. With regular retreatment scheduling, you may not need to think about planning OAB treatment again until your next appointment.
On Screen: If OAB symptoms return before 6 months, talk to your doctor. A minimum of 12 weeks is needed between treatments.
VO: It’s time to ask your doctor if BOTOX® may be the next step in your OAB treatment journey.
Please see Important Safety Information, including Boxed Warning, within the video or on the website below.